Sterilization validation, bioburden, endotoxin, and EO residual testing for medical devices in FDA-registered, ISO 13485-certified facilities.

Sterilization validation is a critical step in medical device manufacturing, ensuring that products meet regulatory requirements, patient safety standards, and market expectations. At Life Science Outsourcing (LSO), we provide comprehensive sterilization validation engineering and testing services, designed to help medical device manufacturers validate, verify, and maintain sterilization effectiveness.
As an FDA-registered, ISO 13485-certified contract manufacturer, our sterilization validation services support compliance with ISO 11135, ISO 17665, and ISO 11737 standards, covering a full range of biological, chemical, and particulate testing.
Bacterial endotoxin testing (BET), or LAL testing, ensures that medical devices and materials meet pyrogen-free requirements to prevent harmful inflammatory responses in patients. LSO provides:


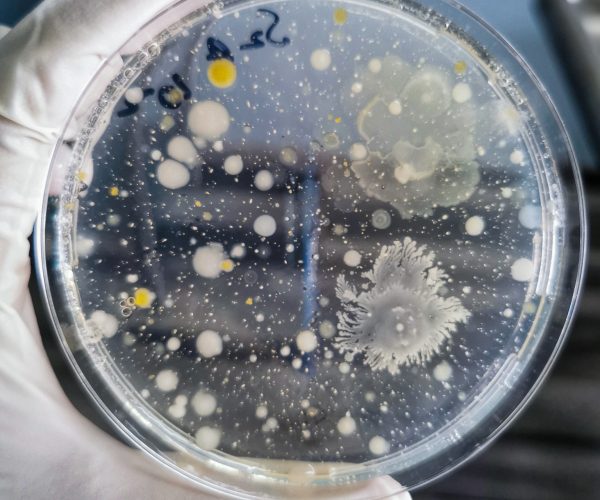
Bioburden testing measures the microbial contamination levels on a medical device before sterilization. Our services include:
Cytotoxicity testing evaluates the potential toxic effects of a medical device on living cells, ensuring biocompatibility and patient safety. LSO offers:
Comprehensive biocompatibility assessments


Regulatory-compliant labeling is critical for traceability, UDI compliance, and patient safety. LSO offers:
Microbial identification is essential for understanding contaminant sources and mitigating risks in medical device manufacturing. Our testing includes:

Particulate contamination can compromise device safety and performance. LSO’s particulate analysis ensures that devices meet USP <788> and ISO 8536-4 standards. Our capabilities include:
Sterility testing confirms that medical devices are free from viable microorganisms after sterilization. Our services include:

Receive the latest infographics, guides, and blog updates for medical device manufacturing, package testing, and sterilization.