Thank you — your request has been received
An LSO specialist will contact you within 1 business day (2 days at most) to discuss your project and next steps.
If your request is urgent, please call us at 714-672-1090.
Why medical device manufacturers choose LSO
• FDA-registered, ISO 13485-certified
• 25+ years supporting Class I, II, and III devices
• In-house assembly, packaging, sterilization, and testing
• Fast project onboarding with regulatory guidance
What to expect next
• We’ll review your project details
• A specialist will reach out by phone or email
• We’ll outline timelines, documentation, and next steps
Explore LSO capabilities
Resources our clients find helpful:

Medical Device Manufacturing
What the FDA’s QMSR Means for Medical Device OEMs and Manufacturing Partners
With FDA’s Quality Management System Regulation taking effect in February 2026, expectations for medical device quality systems are shifting. This Insight examines what is changing under QMSR, why supplier evaluations now look different, and how coherence across quality systems reduces risk for OEMs and their partners.
Medical Package Testing
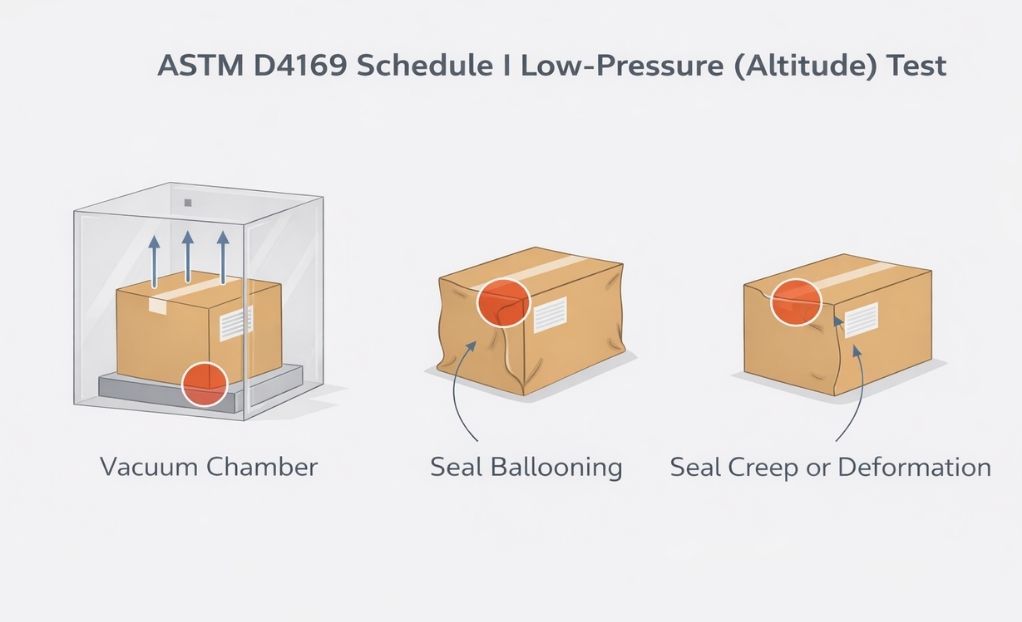
ASTM D4169 Schedule I
Schedule I should be viewed as a pressure-stress lens within ASTM D4169. It does not replace handling, vibration, or compression testing, nor does it independently define distribution robustness.
Its true value emerges when pressure effects are interpreted alongside other hazards to build a defensible, risk-based packaging validation strategy.
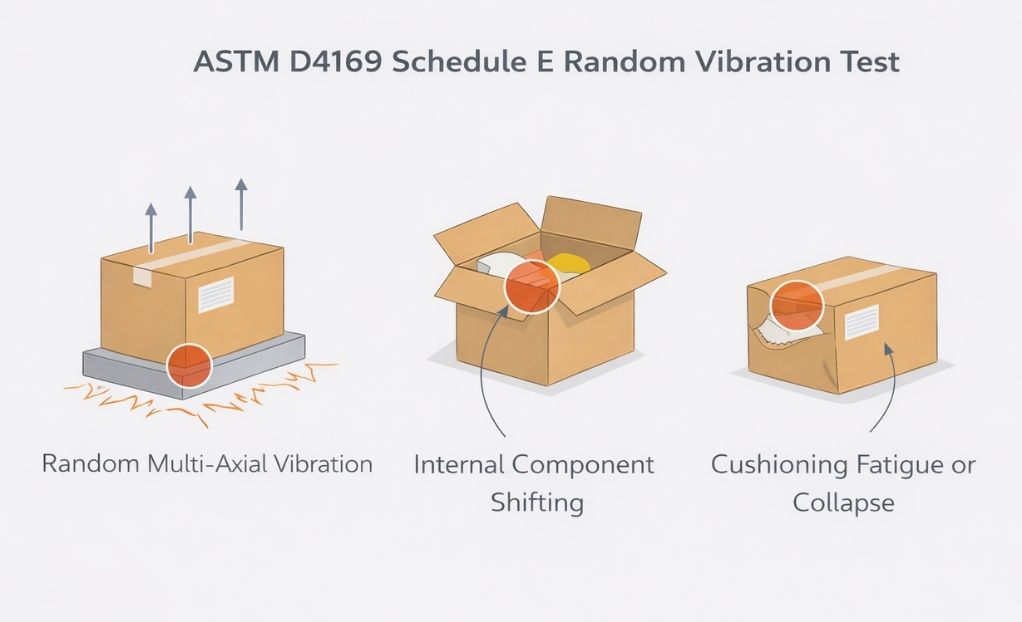
ASTM D4169 Schedule E
ASTM D4169 Schedule E focuses on vehicle vibration realism, exposing packaging systems to sustained, multi-frequency motion that mirrors real-world transportation. Interpreting results requires attention to cumulative effects, assurance level alignment, and subtle indicators of degradation.
When used correctly, Schedule E delivers insight that short-duration or fixed vibration tests simply cannot.
